Single Molecule Electrometer
Single Molecule Electrometer
Just two atoms make the electromechanical sensor reported in the June 29, 2018 issue of Science Advances.[1] Using a single carbon monoxide (CO) molecule attached to the tip of a sharp silver needle, Joonhee Lee and coworkers have demonstrated the world’s smallest voltmeter and have implemented it to measure electric fields inside one molecule (see animation and KUCI radio show "Ask A Leader: Inquiing minds wanna hear" with Claudia Shambaugh).
They monitor the frequency at which the bond between the carbon and oxygen atom of CO oscillates as the test sample is scanned underneath it. The frequency depends on the local electric field sensed by the 1.1 Å-long bond, due to the Stark effect (named after Johannes Stark who received the Nobel Prize in 1919 for his discovery). The signal is relayed optically and detected remotely by relying on the Raman effect (named after Venkata Raman, Nobel Prize in 1930 for his discovery), with the silver needle serving as an antenna to boost the single molecule signal by 12 orders of magnitude, to reach detectable levels. The result is a complete single molecule electromechanical sensor (SMEMS), which can be recognized as the ultimate limit in miniaturization of electromechanics. To put it in perspective, the microelectromechanical systems (MEMS) that are currently deployed in technologies such as cell phones, take their name from the micron size scale (1 micron is 1/100th the thickness of hair). The reported SMEMS is 10000 times smaller, and as far as molecules go, two atoms make the smallest unit.
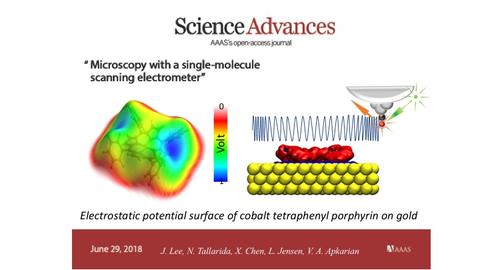
After calibrating the precision electrometer, the researchers used it to map out electric fields inside single molecules. To this end, they used metalloporphyrins, which are ubiquitous in nature and biological systems, from human blood to chlorophyll of plants, and are extensively used display technologies. They discovered that the molecules charge up upon touching gold. The variations in charge distribution within the molecule generates internal fields of order V/Å (100 million Volts per inch…). These are the fields that direct the structural transformations of molecules, i.e., chemistry. The complete description of the fields is color coded on the surface of constant electron density to construct the electrostatic potential surface (EPS), which is the molecular shape of greatest functional relevance (see Figure). Up until recently, the EPS was a theoretical construct, particularly useful in the description of macromolecules such as proteins. The experimentally determined EPS was found to be in good agreement with theory, in the joint experiment-theory study which was published.
Beyond investigations of the molecular world with unprecedented detail, SMEMS can be expected to become the workhorse of technologies such as molecular electronics, and the future of miniaturized devices.
Movie: The CO vibration is excited with a green laser and the red Raman light, which is modulated at the molecular vibrational frequency, is remotely detected. The vibrational frequency of the CO bond changes in response to the sensed electric field. The measured electric fields within a single porphyrin molecules are summarized by the electrostatic potential surface (left). The amplitude of the CO vibrational oscillation is 0.034 Å (100 times smaller than the size of the atom), and its period of oscillation is 16 femtoseconds (fs = 10-15 s).
[1] Microscopy with a Single Molecule Scanning Electrometer, J. Lee, N. Tallarida, X. Chen, L. Jensen, V. A. Apkarian, Science Advances, June, 2018, http://advances.sciencemag.org/content/4/6/eaat5472